|
3/1/2024 0 Comments Potassium and water reaction
The cookies is used to store the user consent for the cookies in the category "Necessary". This cookie is set by GDPR Cookie Consent plugin. The cookie is set by GDPR cookie consent to record the user consent for the cookies in the category "Functional". The cookie is used to store the user consent for the cookies in the category "Analytics". These cookies ensure basic functionalities and security features of the website, anonymously. Necessary cookies are absolutely essential for the website to function properly. Is potassium dissolving in water a chemical or physical change? When you put water on a magnesium fire it causes the water to release hydrogen, which ignites and make the fire burn hotter. What happens when you put water on a magnesium fire? Okay so, the equation for the reaction between potassium and water is the following: So this means that, for every two atoms of potassium and couple molecules of water, we get two molecules of potassium hydroxide (KOH) and one molecule of hydrogen. The Potassium will React VIOLENTLY with the Oxygen in the Water to Produce Potassium Oxide and the Hydrogen will Stick to the Potassium Lump, usually a SMALL One because a Large One would EXPLODE, thus Making the Lump of Potassium Lighter than Water so it Floats !! What happens when you drop potassium in water? Slow reaction with cold water, vigorous with hot water Reactivity series of metals Order of reactivity What products will I get if I add potassium metal to water? What metal reacts with water? It is the seventh most abundant element in Earth’s crust, constituting 2.6 percent of its mass. Potassium imparts a lavender colour to a flame, and its vapour is green. Potassium metal is soft and white with a silvery lustre, has a low melting point, and is a good conductor of heat and electricity. Potassium metal reacts with water to give potassium hydroxide and hydrogen gas. Reaction of potassium with water Potassium metal reacts very rapidly with water to form a colourless solution of potassium hydroxide (KOH) and hydrogen gas (H2). What is the chemical reaction between potassium and water? The metal is also set on fire, with sparks and a lilac flame. It moves around very quickly on the surface of the water. When potassium is added to water, the metal melts and floats. What happens when potassium metal is dropped in water? 3 What products will I get if I add potassium metal to water?. 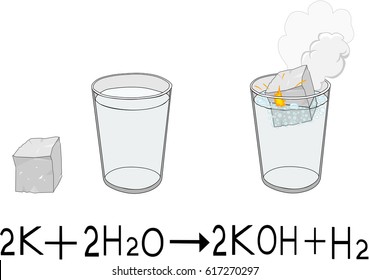
2 What is the chemical reaction between potassium and water?.1 What happens when potassium metal is dropped in water?.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
